Melanoma in childhood and adolescence is under-studied, lacking adequate preventive, diagnostic, and therapeutic strategies. The incidence of melanoma is reported to be about 1.3-1.6 per million in children under 15 years of age and 15 per million in 15-19 y.o., with increasing incidence in adolescents by 4.1% annually since 1997. Additionally, melanoma is among the most frequent solid tumors diagnosed in young adults, having in Europe an incidence of 6.6 per 100.000 and a mortality of 4 per million. Little is known about the interaction between genetic and environmental factors associated with melanoma risk in this context or in the progression from benign nevus to melanoma. Furthermore, lower sensitivity than in adults results in late diagnosis with poorer outcomes, when CAYA patients, particularly the children, could show a 90% 10-year survival rate from stage I/II disease. Thus, melanomas in children, adolescents and young adults (CAYA) represent a medical challenge.
IDENTIFY environmental and genetic risk factors for melanoma in CAYA.
ESTABLISH the molecular profiles of progression from benign congenital nevi to melanoma.
FACILITATE better international collaboration, standards, and melanoma taxonomy.
EVALUATE the clinical efficacy and safety of anti-PD1 antibodies in CAYA melanoma patients
DEVELOP an AI-based diagnostic tool to distinguish the images of melanomas from images of nevi or other benign pigmented skin lesions.
CONDUCT rapid and non-invasive tools for risk and prognosis of melanoma in CAYA.
DESIGN health care system strategies on prevention and diagnosis of melanoma in CAYA and disseminate the results to ensure maximum impact and the widest possible coverage.
ENSURE patient research engagement and education throughout the project and its Implementation
Duration
12/2022 to 11/2026
Programme
Horizon Europe
HORIZON-MISS-2021-CANCER-02
Research & Innovation Action
Reference
101096667
Improving early detection of melanoma in children, adolescents, and young adults
Healthcare professionals will use developed digital tools for risk prediction, early diagnosis and prognosis of melanoma in CAYA.


Putting childhood cancer under the spotlight
Children and adolescents will feel less uncomfortable and scared in medical visits due to the use of volatilities detection through skin and breath or liquid biopsy.
Making the most of data and digitalisation in cancer prevention and care
MELCAYA’s second opinion platform will become a standard for melanoma diagnosis in CAYA in Europe and beyond. Additionally, anonymized melanoma and L/GCMN images collected in MELCAYA will be used by other hospitals, researchers and innovators for further research


Building on the promise of personalised medicine for cancer prevention, diagnosis and treatment
Health care professionals will use risk scores and
digital platform developed in MELCAYA in order to improve the personalization of L/GCMN and melanoma cases in CAYA.
Reducing cancer inequalities across the EU
MELCAYA digital platforms and tools will be validated and used in 5 countries and 50 patients
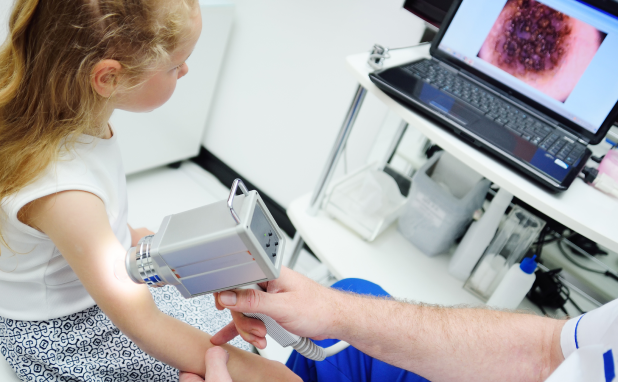
WP1 aims to (1) identify risk factors involved in the development of melanoma in CAYA, including patients with L/GCMN using registry data; (2) characterize the exposome in CAYA with melanoma, including patients with L/GCMN; (3) enumerate molecular determinants of personal risk from endogenous or exogenous sources, and (4) test pre-clinical strategies to best model and thereby improve patient response.
WP2 aims to (1) identify the molecular profiles of all cellular states of progression from benign nevi to malignant melanoma in young patients with congenital predispositions, (2) longitudinally characterize the cell-free DNA (cfDNA) from young L/GCMN and melanoma patients, (3) improve early diagnosis, prevention and treatments for intermediate conditions such as large/giant congenital melanocytic nevi (L/GCMN) through evidence-based interpretation of personal risk from endogenous or exogenous sources, and (4) test pre-clinical strategies to best model and thereby improve patient response.
WP3 seeks to (1) identify tumor tissue processing and quality control protocols, (2) develop a novel hybrid taxonomy (3) create a pan-European second opinion platform for collaboration between pathologist, and (4) facilitate European standardization and knowledge sharing of clinically validated biomarkers, tools and algorithms
WP4 aims to (1) retrieve, in the early stage and metastatic disease, data on the activity and efficacy of anti PD1 antibodies administered to childhood, adolescent and young adult melanoma patients in the context of academic European centers, (2) evaluate acute and long-term toxicity of anti PD-1 antibodies in adolescent, childhood and young adult with early and advanced melanomas, (3) investigate in childhood, adolescent and young adult
melanoma population, the impact of anti PD-1 antibodies on fertility, and (4) evaluate the incidence of second cancers in CAYA with early and advanced melanomas treated with anti PD-1 therapy.
WP5 seeks to (1) to generate AI-based image analysis classifiers as melanoma risk prediction biomarkers and diagnostic assistance tools for CAYA melanoma early detection. The project will contribute the data that will be used to train and test the classifiers. Participating dermatologists and pathologists will contribute to data annotation and analyze the results of the XAI-based algorithms.
WP6 aims to (1) develop an AI-based solution for the quantitative prognosis of melanoma in CAYA, (2) implement an interpretability module of the outputs of the model, (3) create a new statistically coherent stratification and policy for melanoma in CAYA, (4) design, produce, and test novel non-invasive sensing technologies.
WP7 aims to (1) develop guidance on the assessment of innovative technologies addressed to the prevention and diagnosis of melanoma in childhood, including advice on their transferability to EU health system under the current EU Health Technology Assessment Regulation, (2) assess the ethical, legal and social implications (ELSI) on implementing preventive and diagnostic health care schemes for melanoma in childhood, (3) design health care system strategies to inform policy decisions to be implemented in overcoming current gaps in EU markets on prevention and diagnosis of melanoma in childhood.
WP8 seeks to manage and implement effective, strategic communication, engagement and exploitation activities with the aim to increase awareness, acceptance and uptake of MELCAYA results at national, European and global levels.
WP9 aims to (1) ensure the engagement of European patient organizationsin the MELCAYA research and education of patients and patients advocates and at the same time assuring its results align with the melanoma patient community expectations and giving the social and humanities perspective to the project, (2) to facilitate regular contact between the melanoma patient community and consortium partners to ensure mutual learning, (3) to assure that conducting ethnographic research within our patient community, (4) to coordinate the involvement of the wider European melanoma community via the Melanoma Patient Network Europe (MPNE), including patients from CEE via Melanoma Patient Network in Central and Eastern Europe (MPN-CEE).
WP10 aims to ensure efficient administrative and financial management, to coordinate the MELCAYA project by monitoring the successful implementation of the different research activities within the agreed time and cost, through continuous quality assessment and monitoring of potential risks and their appropriate mitigation, to guarantee the implementation of the Grant Agreement and the Consortium Agreement, to deliver timely reporting on project progress, to manage the data generated, collected and processed by the project, to handle relevant ethical issues, as well as the open access of publications.
WP11 aims to ensure compliance with the ‘ethics requirements’ set out in this work package.
D1.1 Risk factors and exposome for melanomas in CAYA
D1.5 ExpoMel initiation package
D1.6 ExpoMel Midterm recruitment report
D1.7 ExpoMel Report on the status of posting results in a repository
D2.1 Partner registration status
D2.2 Report on molecular data collection of L/GCMN
D2.4 Mid-term report on quality control
D2.6 NevustoMel initiation package
D2.7 NevustoMel Midterm recruitment report
D2.8 NevustoMel Report on the status of posting results in a repository
D3.1 Standardized protocols for tumor tissue processing
D3.2 Standardized protocols and procedures for melanoma sub-classification
D3.5 Mol-Mel initiation package
D3.7 Mol-Mel Report on the status of posting results in a repository
D4.2 Immuno-Ped initiation package
D4.3 Immuno-Ped Midterm recruitment report
D4.4 Immuno-Ped Report on the status of posting results in a repository
D4.5 Immuno-Ped II initiation package
D4.6 Immuno-Ped II Midterm recruitment report
D4.7 Immuno-Ped II Report on the status of posting results in a repository
D5.4 AI-MEL initiation package
D5.5 AI-MEL Midterm recruitment report
D5.6 AI-MEL Report on the status of posting results in a repository
D6.4 Precis-mel initiation package
D6.5 Precis-mel Midterm recruitment report
D6.6 Precis-mel Report on the status of posting results in a repository
D7.1 Framework for assessing innovative HTs
D7.2 Set of Health care system strategies
D8.1 Dissemination & communication plan 1
D8.2 Dissemination & communication plan 2
D8.3 Dissemination & communication plan 3
D8.4 Exploitation Strategy
D8.5 Online and media presence 1
D8.6 Online and media presence 2
D8.7 Engagement summary report
D8.8 Project Website
D8.9 Common work plan for scientific collaboration under the ‘Understanding’ cluster
D8.10 Common video and/or a common cluster brochure
D9.1 Patient engagement plan
D9.2 Impact assessment of patient engagement
D9.3 Report on the methodology for ethnographic citizen science to understand barriers in CAYA patients
D9.4 Citizen engagement summary report
D9.5 Addressing inequalities recommendations
D10.1 Project Management Plan
D10.2 Quality Assurance and Risk Management Plan
D10.3 Data Management Plan 1
D10.4 Data Management Plan 2
D10.5 Data Management Plan 3
D10.6 Plan for ethic-legal monitoring & ethical submissions 1
D10.7 Plan for ethic-legal monitoring & ethical submissions 2
D10.8 Conclusions of common annual meeting of the ‘Understanding’ cluster 1
D10.9 Conclusions of common annual meeting of the ‘Understanding’ cluster 2
D10.10 Conclusions of common annual meeting of the ‘Understanding’ cluster 3
D10.11 Conclusions of common annual meeting of the ‘Understanding’ cluster 4
D10.12 Policy brief formulating recommendations based on the research and innovation strand of the ‘Understanding’ annual cluster meeting 1
D10.13 Policy brief formulating recommendations based on the research and innovation strand of the ‘Understanding’ annual cluster meeting 2
D10.14 Policy brief formulating recommendations based on the research and innovation strand of the ‘Understanding’ annual cluster meeting 3
D10.15 Policy brief formulating recommendations based on the research and innovation strand of the ‘Understanding’ annual cluster meeting 4

PERSONALIZED PLANS
FOR EVERYONE
Cu primis bonorum recteque vis, vidit conclusionemque cum ei, eu accusam assentior. Affert ubique molestiae at sit, scripta ancillae vix ad neglegentur.
- Ne numquam senserit democritum qui
- Error scripta equidem vim ea.
- Dicant graece principes te vel, pro voluptua
- Sit ea malis munere causae
- Atqui percipitur et cum, vix consequat dissentiet
’’Turpis egestas pretium aenean pharetra. Facilisi etiam dignissim diam quis enim lobortis scelerisque. Nullam non nisi est sit. Sapien faucibus et molestie ac feugiat sed.’’
rick greenwalt - fitness instructor
’’Et egestas quis ipsum sendisse ultrices gravida dictum. Placerat vestibulum lectus mauris ultrices. Porta non pulvinar neque laoreet suspendisse interdum consectetur.’’
Bernard Curtis - Cardio Instructor
’’Dictum non consectetur a erat nam. Risus viverra adipiscing at in tellus. Cursus metus aliquam eleifend mi in nulla posuere. Viverra maecenas accumsan lacus.’
Mae Kurt - Cardio Instructor
No posts were found for provided query parameters.

